Duke University Researchers Find Body's Heat Sensors - Cause of Pain Revealed
A research team from the Duke University has recently discovered a protein (TRPV2 polymodal heat sensor), which is directly linked to pain and heat perception in bodies of animals. It is basically an ion channel in the cell surface membrane dubbed as TRPV2. These pain receptors play a vital role in biological processes, such as maintaining a healthy heart, helping in disposal of pathogens and influence cell death (apoptosis) in cancer.
The body mechanism relies on heat sensors and pain receptors, in order to protect the body from severe burning and to detect any injury or complication in the body respectively. These tiny biological sensors comprehend the danger and send the information to brain to respond.
The research team from DU, developed a blue print of a temperature sensitive Transient receptor potential vanilloid 2(TRPV2,a family member of TRPV) using the Cryo-electronic microscopy technique. Senior study author Seok-Yong Lee, an assistant professor of biochemistry at the Duke University School of Medicine, explained that these sensors had tricked human beings for several years due to their complicated structure. He claimed that their research on TRPV2’s work mechanism would revolutionize contemporary sensation therapeutics.
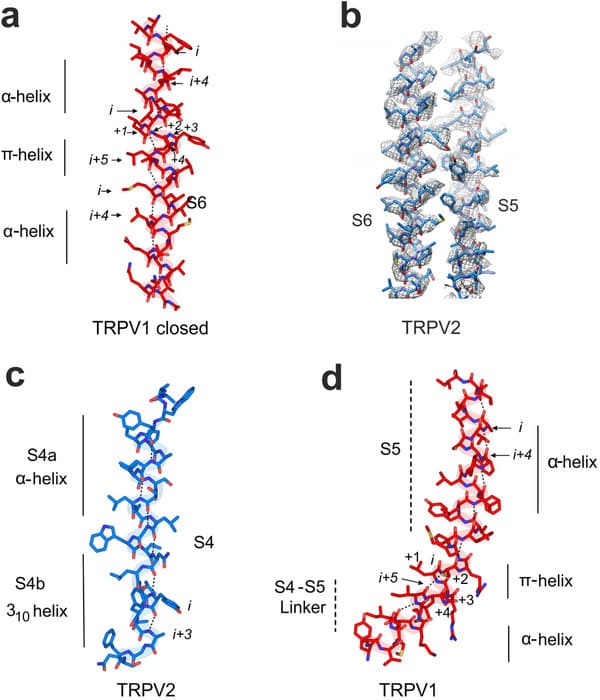
TRPV2 And TRPV1
Ion channels are scattered across all cell membranes, among which the TRPV2 uses Calcium ions to indicate intimacy of the heat energy source with the body. The TRPV2 works like a control valve, where heat or other stimuli can open the valve and allow a surge of calcium ions to flow towards the brain. But, till now people have been in the dark regarding the exact working mechanism of the TRPV2 receptors.
Recently, the blue print of the TRPV1 structure was discovered, which refurbished the making of efficient opioids (popularly known as painkillers). Unlike TRPV1 which is only found in the nervous system, TRPV2 is present throughout the body and has been observed in a variety of human conditions, including heart disease, the immune response, and cancer. However imaging the structure was much harder than understanding the underlying concept.
Gabriel Lander, a biologist at the Scripps Research Institute in La Jolla, Calif used cryo-electron microscopy to generate half a million 2D images of frozen TRPV2, which was then subsequently processed to create a 3D model of the TRPV2 structure. The resultant image however contradicted with the TRPV1 open or closed structure leading to the confirmation of another 3rd state, between the open and closed channels.
Lee explained that their future plan is to create a series of snapshots of the TRPV2 structure that might reveal the working principle of these polymodal sensors. The research was supported by the Duke University Medical Center and the National Institutes of Health (R01GM100894, DP2OD008380, and DP2EB020402), the Searle Scholars Program, and the Pew Charitable Trusts. The research was published in the Nature Structural and molecular biology journal.
Source: <a href="https://today.duke.edu/2016/01/painsensor?utm_source=feedburner&utm_medium=feed&utm_campaign=Feed%3A+duke%2FaSRA+%28Research%29" target="_blank" rel="nofollow noopener noreferrer">Scientists Discover Blueprint of Body's Temperature Sensor | Duke Today</a> | #-Link-Snipped-#
The body mechanism relies on heat sensors and pain receptors, in order to protect the body from severe burning and to detect any injury or complication in the body respectively. These tiny biological sensors comprehend the danger and send the information to brain to respond.
The research team from DU, developed a blue print of a temperature sensitive Transient receptor potential vanilloid 2(TRPV2,a family member of TRPV) using the Cryo-electronic microscopy technique. Senior study author Seok-Yong Lee, an assistant professor of biochemistry at the Duke University School of Medicine, explained that these sensors had tricked human beings for several years due to their complicated structure. He claimed that their research on TRPV2’s work mechanism would revolutionize contemporary sensation therapeutics.

TRPV2 And TRPV1
Recently, the blue print of the TRPV1 structure was discovered, which refurbished the making of efficient opioids (popularly known as painkillers). Unlike TRPV1 which is only found in the nervous system, TRPV2 is present throughout the body and has been observed in a variety of human conditions, including heart disease, the immune response, and cancer. However imaging the structure was much harder than understanding the underlying concept.
Gabriel Lander, a biologist at the Scripps Research Institute in La Jolla, Calif used cryo-electron microscopy to generate half a million 2D images of frozen TRPV2, which was then subsequently processed to create a 3D model of the TRPV2 structure. The resultant image however contradicted with the TRPV1 open or closed structure leading to the confirmation of another 3rd state, between the open and closed channels.
Lee explained that their future plan is to create a series of snapshots of the TRPV2 structure that might reveal the working principle of these polymodal sensors. The research was supported by the Duke University Medical Center and the National Institutes of Health (R01GM100894, DP2OD008380, and DP2EB020402), the Searle Scholars Program, and the Pew Charitable Trusts. The research was published in the Nature Structural and molecular biology journal.
Source: <a href="https://today.duke.edu/2016/01/painsensor?utm_source=feedburner&utm_medium=feed&utm_campaign=Feed%3A+duke%2FaSRA+%28Research%29" target="_blank" rel="nofollow noopener noreferrer">Scientists Discover Blueprint of Body's Temperature Sensor | Duke Today</a> | #-Link-Snipped-#
0
