Molybdate, a new catalyst can turn CO2 emissions into useful polymers
Engineers at MIT have discovered a new catalyst called Molybdate (one molybdenum atom bonded with four oxygen atoms) that offers the much needed capability of trapping the carbon dioxide in air and converting it into useful polymers. The research is at primary stage and a lot more work is needed before efficient and economical processes can be developed to trap CO2; but if successful - it can help humans control the global warming to a greater extent. Christopher Cummins, professor of Chemistry at MIT says that Molybdate has the ability to fix the CO2 emitted by automobiles, factories and power plants - which continue to be the among the top sources of CO2. The aim behind the research is to develop carbon-neutral cycles for the cleaner energy and also minimize the pollution levels.
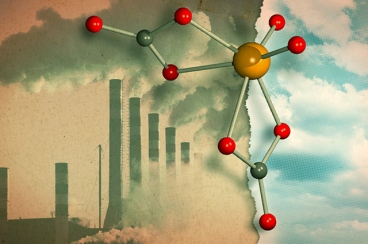
Credits: JOSE-LUIS OLIVARES/MIT; MOLYBDATE 3-D RENDERING BY LOANA KNOPF
The new reaction developed by chemists turns CO2 into carbonate ion (negatively charged) which reacts with silicon compound to create formate. Formate is one of the common starting material used in manufacturing of organic compounds. The process relies on molybdate - a catalyst that may be employed for larger scale industrial application. Molybdate is available abundantly and is stable in both air and water. No efforts were made until now to figure out how molybdate interacts with CO2.
Though Cummins and team have found promising results; they believe more research is needed to make the process useful for industries. They're now working on figuring out ways to trigger the reaction that results into creation of molybdate - which can be further used to catalyse other reactions.
The process of converting CO2 into various useful organic compounds is termed as 'Carbon Fixation'. The challenge lies in making carbon fixation economical and ready for mass adoption by the industries so that greater control can be achieved over the levels of CO2 in air. We wish Cummins and team all the best. Those interested in reading more about the research may head over to the source link below.
Source: #-Link-Snipped-#

Credits: JOSE-LUIS OLIVARES/MIT; MOLYBDATE 3-D RENDERING BY LOANA KNOPF
The new reaction developed by chemists turns CO2 into carbonate ion (negatively charged) which reacts with silicon compound to create formate. Formate is one of the common starting material used in manufacturing of organic compounds. The process relies on molybdate - a catalyst that may be employed for larger scale industrial application. Molybdate is available abundantly and is stable in both air and water. No efforts were made until now to figure out how molybdate interacts with CO2.
Though Cummins and team have found promising results; they believe more research is needed to make the process useful for industries. They're now working on figuring out ways to trigger the reaction that results into creation of molybdate - which can be further used to catalyse other reactions.
The process of converting CO2 into various useful organic compounds is termed as 'Carbon Fixation'. The challenge lies in making carbon fixation economical and ready for mass adoption by the industries so that greater control can be achieved over the levels of CO2 in air. We wish Cummins and team all the best. Those interested in reading more about the research may head over to the source link below.
Source: #-Link-Snipped-#
Replies
-
 Joseph ChristieWe all remember the Ammonium phosphomolybdate reaction in qualitative inorganic chemistry for phosphates. Aqueous solution of Ammonium Molybdate gives a bright yellow phosphomolybdate precipitate. In agricultural chemistry this reaction is used to quantify PO4 (-3) in soil or fertilizer mixtures.
Joseph ChristieWe all remember the Ammonium phosphomolybdate reaction in qualitative inorganic chemistry for phosphates. Aqueous solution of Ammonium Molybdate gives a bright yellow phosphomolybdate precipitate. In agricultural chemistry this reaction is used to quantify PO4 (-3) in soil or fertilizer mixtures.
It is intriguing that the MoO4 (-2) , similar to sulfate Ions in structural symmetry, has the ability to " to fix the CO2" as reported by the MIT researchers,. The reaction path involving the conversion of CO2 to CO3 (-2) and the eventual formation of formate ion , HCOO-, the basic unit or precursor to the synthesis of other compounds, catalyzed by MoO4 (-2) is fascinating chemistry.
I hope the MIT workers will soon come up with promising and positive results.
You are reading an archived discussion.
Related Posts
People believe WhatsApp free and while I'm using it for quite some time now - let me tell you; it's not free. It's cheaper than SMS; but it's *NOT FREE*....
Engineers from M S Ramaiah Institute of Technology, Bangalore have come with a new free voice calling service called 'FreeKall'. Developed by a trio of Yashas Shekar, Vijayakumar Umaluti and...
Name: Er. B C Dinesh
*Engineering Trade: CIVIL Engineering
Location: MAngalore, Karnataka
*Occupation: Working
Work Experience: 22+ Years
*Hobbies & Interests: Interested in Innovative Engineering, Expertise In Cable stayed and...
Gaurav Dahake submitted a new project:
BuyHatke Browser Add On For Google Chrome & Mozilla Firefox - A revolutionary hack for getting the best price always
I am an engineer...
Developed by Robert Jacob, a computer scientist and Sergio Fantini, a Biomedical engineer from Tufts University’s Human-Computer Interaction Lab, a new headband that will read brain's activity and use this...
